On-Campus Research Moves to Phase 2
October 12, 2020
Dear Teachers College Research Community –
Now that the fall semester is thoroughly underway, we want to update you on progress towards resuming in-person human subjects research. As a reminder:
Due to COVID-19, in-person research data collection activities on the Teachers College campus, and at remote sites by Teachers College personnel, are suspended.
We are writing as Co-Chairs of the Research Compliance & Safety Committee, formerly the Research Ramp-Up Task Force. The goal of the committee is to harness content-specific expertise, initially to establish a plan for in-person research ramp-up at Teachers College with different phases that permit occupancy to be scaled depending on the scenario, personnel, and study site. Each phase should be adaptable as appropriate for the circumstances pertaining to specific research groups and risk levels.
The committee’s continuing mandate beyond the pandemic is to evaluate, establish and maintain aspects of regulatory compliance, risk management, and safety for researchers and research participants in the Teachers College community.

The Research Compliance & Safety Committee has served as a guide to the Institutional Review Board (IRB) on research-related matters and human subjects protection strategies in the time of COVID-19. We recently launched research-related information on TC’s Preparedness website including a four-phase process for returning to in-person research activities. We have been in Phase 1 for the initial parts of the semester while the College reopening procedures were established.
As of now, we are moving to Phase 2.

This means that Faculty Researchers and Research Staff, including students who work in research, may access on-campus labs and research facilities provided that all campus safety requirements are met.
Requirements for moving into Phase 2 are detailed here.
And include:
- Teachers College Environmental Health and Safety Training
- Daily completion of self-health check
- Recent negative COVID-19 test
Teachers College is implementing a 0% to 25% on-site employee model to achieve physical distancing in the workplace by maintaining remote operations for non-essential employees and office activities. Researchers returning to campus must adhere to this on-site campus model.
We are all aware that some research studies cannot sustain long-term in-person data collection suspensions. We anticipate being able to move to Phases 3-4 over the next several months and we are already working with individual research labs and centers to support this gradual transition.
Over the coming weeks, we will send out more information about requirements for resuming in-person data collection on-and-off campus—including the “Ethics Amid Uncertainty Research-Related Training Modules,” geared for researchers who will conduct in-person data collection. We will make an announcement when the link to these training modules is available on the Research Compliance & Safety website.
We aim to support researchers through the many hurdles in preparation for eventual in-person data collection. Changes or guidelines to in-person data collection will follow an iterative process. Each phase will require a feedback period as we refine and scale-up researcher guidelines. In other words, no changes will be made in haste and your patience through this process is essential. We are taking care to weigh decisions and assess risks for both researchers and research participants. In addition to our Committee, we are also consulting with campus staff and researchers familiar with safety and public health.
As a reminder, the procedures to become eligible to resume in-person data collection with human participants are threefold:
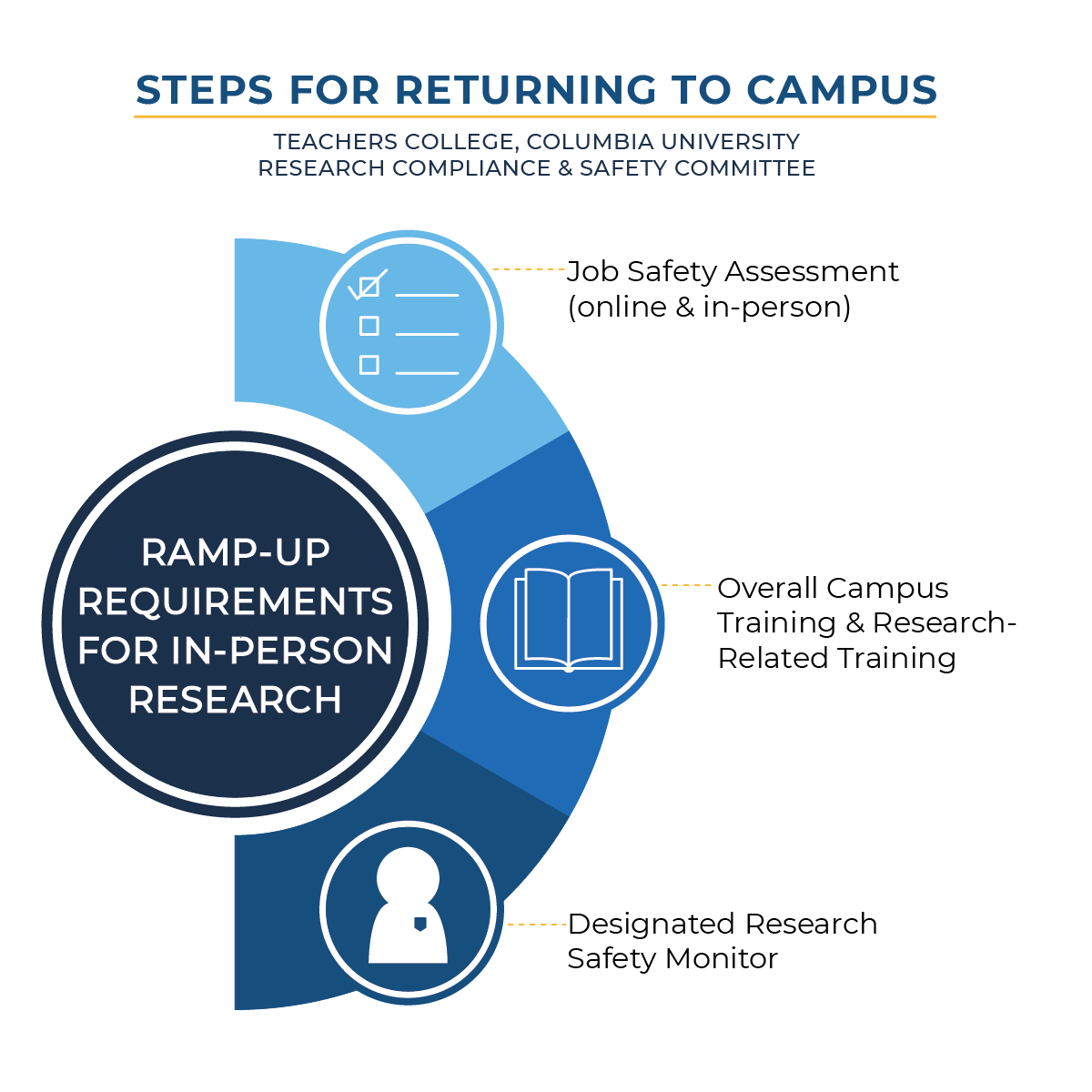
- Job Safety Assessments have been conducted with the Offices of Environmental Health and Safety and Facilities, and reports will soon go out to individual researchers detailing safety requirements determined as a result of those assessments. Please contact Patrick Mathelier, Director of Environmental Health and Safety (ehs@tc.columbia.edu) if you wish to schedule a Job Safety Assessment (JSA).
- The Teachers College Environmental Health and Safety Training is already available and is required in order to enter campus in any capacity. The “Ethics Amid Uncertainty Research-Related Training Modules,” is under development and will soon be available; this is required before researchers may resume in-person data collection activities.
- The role of a designated Research Safety Monitor (RSM) requires completion of the Job Safety Assessment and the “Ethics Amid Uncertainty Research-Related Training Modules.” The RSM will also be asked to complete an agreement to adhere to a hygiene and safety policy within their own research lab or center.
The Teachers College Institutional Review Board (TC IRB) will maintain oversight of research involving human subjects, and resuming in-person data collection will require IRB approval and the completion of these three steps.
We do understand that some research may be deemed essential, may carry direct benefits to participants, or may need to adhere to strict obligations or deadlines. If there are specific, extenuating circumstances that you believe constitute grounds for resuming in-person study activities ahead of the phased plan, please indicate this information in Mentor IRB for review and determination.
- You can do this by navigating to your protocol page in Mentor IRB. Click on the Modifications tab and select “Create New Modification.” Check the box next to "My Study Methods Cannot be Moved Online for COVID-19 and Study Cannot be Paused" and then, when the modification is created, click on the link to the "My Study Cannot be Moved Online" (links in Mentor IRB usually appear as dark red text). This step will take you to a short survey to provide more information as to why your study should include direct face-to-face interaction with a research participant. TC IRB will make the final determination on your modification submission.
- Please review the How to Submit COVID-19 Modification Studies Not Online walkthrough guide for studies that cannot moved online.
- We also have a walkthrough guide for instances when some of your research activities must remain in-person, yet others can be moved online. In these cases, please review the How to Submit COVID-19 Modifications for Hybrid Activities walkthrough guide.
Resuming in-person data collection with human participants will be dependent on continued monitoring of the health and safety of our community, and on approvals from TC IRB, the Offices of the Provost, and Environmental Health and Safety.
In the next few weeks, we will also post new researcher materials:
- A new External Site Permission form that includes safety and hygiene language
- Revised consent, permission, assent forms which include statements about:
- Person-to-person exposure is the most frequent route of transmission for infectious viruses and occurs via direct inhalation of respiratory droplets during close contact.
- Ways to mitigate risk (social distance, wear face covering).
- (Limited) Mandated Reporting: When required by law, information (including individually identifiable information) related to a research subject’s COVID-19 tests results may be reported to a public health authority.
We ask you all to continue to work remotely (online) for as long as possible. Minimizing the density of personnel on campus is crucial for everyone’s safety. Please do not engage in the return to research activities on campus unless this is crucial for your work. We will gladly help if you wish to have a discussion in order to make that determination. Please feel free to reach out by email with any comments, questions, or concerns.
If you would like to contribute your insights to the design of our procedures and policies, you can:
- Complete this confidential 15-minute survey.
- Volunteer for a 15-minute, semi-structured interview (email IRB@tc.edu, with a subject line of “Research Input for Research Ramp-Up”)
As ever, we are working hard to ensure everyone’s safety as we cautiously resume aspects of our research operations. We remain grateful for your flexibility and patience, and wish you all continued health, safety, and productivity.
Best wishes –

Karen Froud, Ph.D., IRB Chair
Myra Luna-Lucero, Ed.D., Director of Research Compliance
